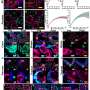
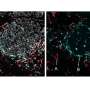
Junctional length establishes the onset of tissue plasticity. a, Sketch showing a lateral view of a ten-somite stage embryo highlighting the posterior region of the body (dotted black rectangle) where mesodermal progenitors progressively differentiate as they transit from the MPZ to the PSM (i). Confocal sections along the sagittal plane of posterior extending tissues in membrane-labeled Tg(actb2:MA-Citrine) embryos (inverted) containing ferrofluid droplets (cyan) in different regions along the AP axis, namely the A-PSM, the posterior PSM (P-PSM) and the MPZ (ii). V, ventral; D, dorsal; A, anterior; P, posterior. The red dashed contours highlight each region (A-PSM, P-PSM and MPZ). b, Confocal section of droplet (cyan) during actuation (magnetic field ON; membrane label, inverted; i). Red dashed line indicates droplet contour and arrows indicate the direction of forces applied by the droplet. Sketches defining the induced droplet elongation e along the direction of the applied magnetic field HH and the droplet pre-elongation before actuation, e0 (ii). Dashed lines indicate the unelongated droplet. c, Mean (dots) and median (lines) values of cell size (diameter, d; gray) and junctional length (ℓ; red) in the MPZ and PSM, reanalyzed from the literature. The inset at the right shows illustrates the parameters. Error bars, s.e.m. d, Examples of the time evolution of droplet deformation (normalized droplet extension, e/d¯, where d¯ is the average cell size (diameter); induced strain) during actuation cycles (OFF–ON–OFF) for different values of the applied magnetic field, leading to varying eM values. Both e0 and eR are defined in the inset. e,f, Residual droplet elongation normalized by average cell size, (eR−e0)/d¯(e) or by average junctional length, (eR−e0)/ℓ¯ (f), for varying values of the applied maximal droplet elongation (eM − e0) normalized by average cell size (e) or junctional length (f) in the posterior paraxial mesoderm. Vertical red lines in e and f indicate the onset of plastic, irreversible deformation. Median and interquartile range are shown. P values were obtained from one-sample two-tailed t-tests. NS, not significant; **P = 0.0025, ****P < 0.0001. g, Snapshots showing confocal sections of tissue next to a droplet (dotted red line) during an actuation cycle (OFF–ON–OFF) causing T1 transitions that lead to plastic changes in the local tissue architecture (bottom). Credit: Nature Materials (2022). DOI: 10.1038/s41563-022-01433-9
Researchers in a variety of disciplines have been fascinated by cell communication for decades. Professor Otger Campàs together with his colleagues from the Physics of Life (PoL) Cluster of Excellence at Technische Universität Dresden and from the University of California Santa Barbara (UCSB) have now been able to unravel another mystery surrounding the question of how cells use their sense of touch to make vital decisions during embryogenesis. Their paper has now been published in the journal Nature Materials.
Testing the surroundings
In their paper, the researchers report how cells within a living embryo mechanically test their environment and what mechanical parameters and structures they perceive. "We know a lot about how cells sense and respond to mechanical cues in a dish. However, their microenvironment is quite different within an embryo and we did not know what mechanical cues they perceive in a living tissue," says Campàs, Chair of Tissue Dynamics and PoL Managing Director.
The mechanical cures helps cells make important decisions, such as whether or not to divide, move or even differentiate, the differentiation process by which stem cells turn into more specialized cells able to perform specific functions.
Previous research revealed that stem cells placed on a synthetic substrate rely heavily on mechanical cues to make decisions: Cells on surfaces with a stiffness similar to bones became osteoblasts (bone cells), whereas cells on surfaces with a stiffness similar to brain tissue became neurons. The findings greatly advanced the field of tissue engineering as researchers used these mechanical cues to create synthetic scaffolds to coax stem cells to develop into desired outcomes. These scaffolds are used today in a variety of biomedical applications.
From a dish to the living embryo
However, a dish is not the cell's natural habitat. While building an organism, cells are not in contact with synthetic scaffolds in a flat dish, but rather with complex living materials in three dimensions.
Over the last decade, Prof. Campàs' research group uncovered the mechanical cues that guide cells in the complex tissues of an embryo. Using a unique technique developed in his lab, the researchers could probe the living tissue in a similar way as cells do and find out what mechanical structures the cells sense.
"We first studied how cells mechanically test their micro-environment as they differentiate and build the body axis of a vertebrate, as they differentiate," Campàs says. "Cells used different protrusions to push and pull on their environment. So we quantified how fast and strong they were pushing."
By using a ferromagnetic oil droplet that they inserted between developing cells and subjecting it to a controlled magnetic field, they were able to mimic these tiny forces and measure the mechanical response of the cells surroundings.
Sensing the tissue architecture and cells change fate
Critical to these embryonic cells' actions is their collective physical state, which Campàs and his research group described in a previous paper to be that of an active foam, similar in consistency to soap suds or beer froth, with cells clumped together by cell adhesion and tugging of each other.
What the cells are mechanically probing, Campàs and team found out, is the collective state of this "living foam"—how stiff it is and how confined the assemblage is. "And right at the moment that cells differentiate and decide to change their fate, there is a change in the material properties of the tissue that they perceive." According to him, at the moment the cells within the tissue decide on their fate, the tissue falls in stiffness.
Going forward
What's not yet proven in this study is the complex question of whether—and if so, how—the change in the stiffness in the embryonic environment drives the change in the cell state.
"There is an interplay between the mechanical characteristics of the structures that cells collectively build, such as tissues or organs, and the decisions they make individually, as these depend on the mechanics cues that cells sense in the tissue. This interplay is at the core of how nature builds organisms," says Campàs.
The findings from this study might also have important implications for tissue engineering. Potential materials that mimic the foam-like characteristics of the embryonic tissue, as opposed to the widely used synthetic polymer or gel scaffolds, may allow researchers to create more robust and sophisticated synthetic tissues, organs and implants in the lab, with the appropriate geometries and mechanical characteristics for the desired functions.
More information: Alessandro Mongera et al, Mechanics of the cellular microenvironment as probed by cells in vivo during zebrafish presomitic mesoderm differentiation, Nature Materials (2022). DOI: 10.1038/s41563-022-01433-9
Journal information: Nature Materials
Provided by Dresden University of Technology